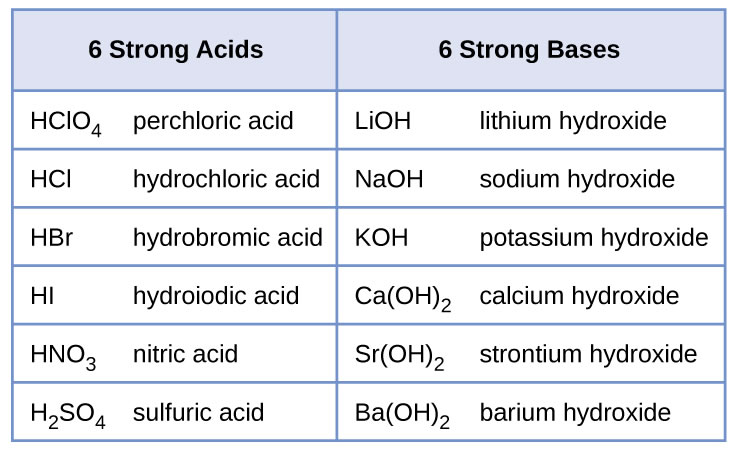
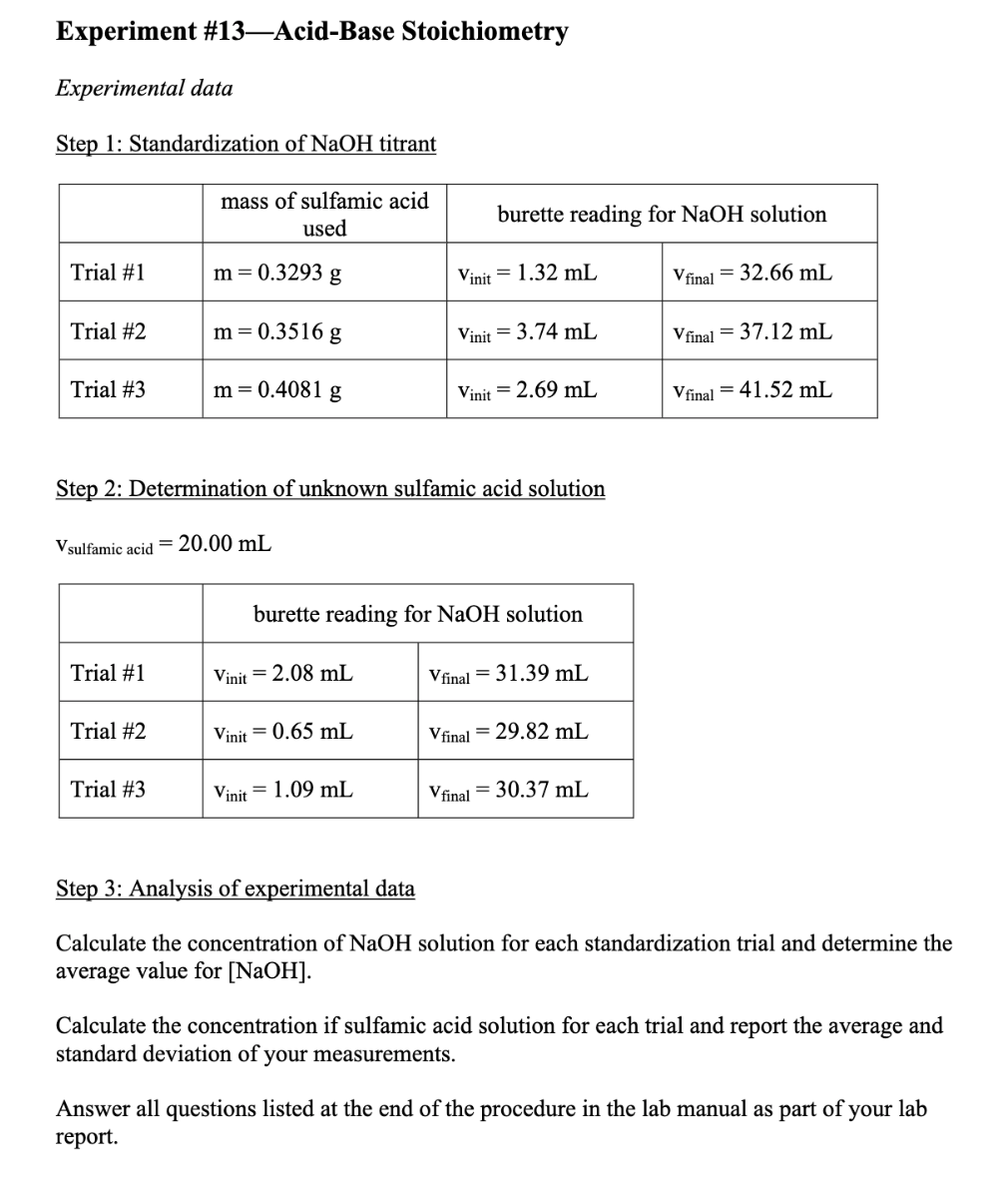
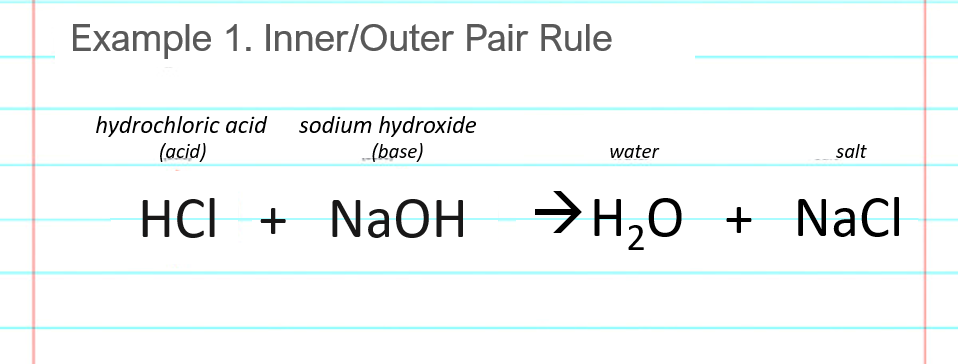
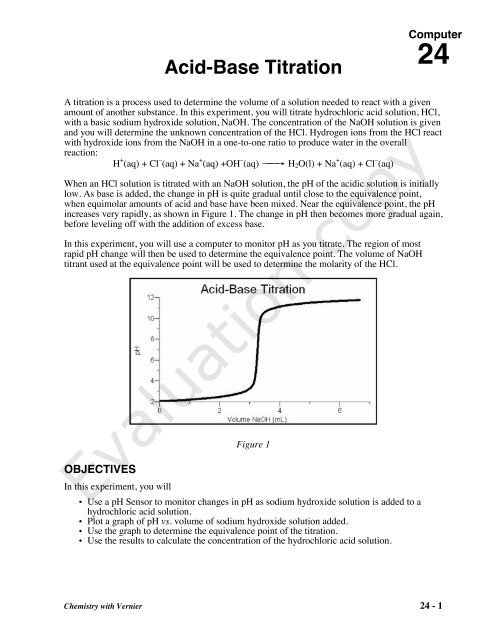
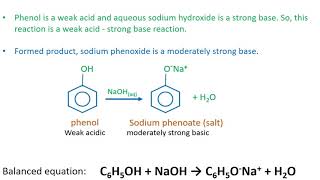
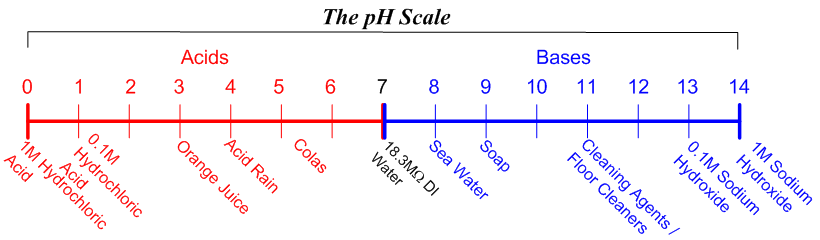
SOLVED: LA REPORT SHEET EXPERIMENT Titration of Acids and Bases 20 A. Analysis of an Unknown Acid Trial 3 Trial 2 Trial Mass of bottle unknown Mass of bottle Mass of unknown
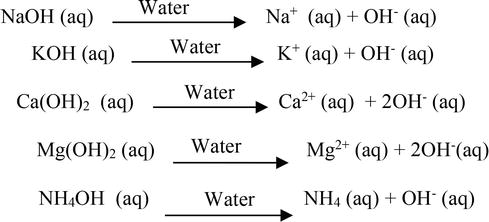
Sodium hydroxide (NaOH) is classified as a strong base. For every mole of sodium hydroxide added to a large volume of water, one mole of what ion enters the solution? | Socratic